Publications |
* = Corresponding Author |
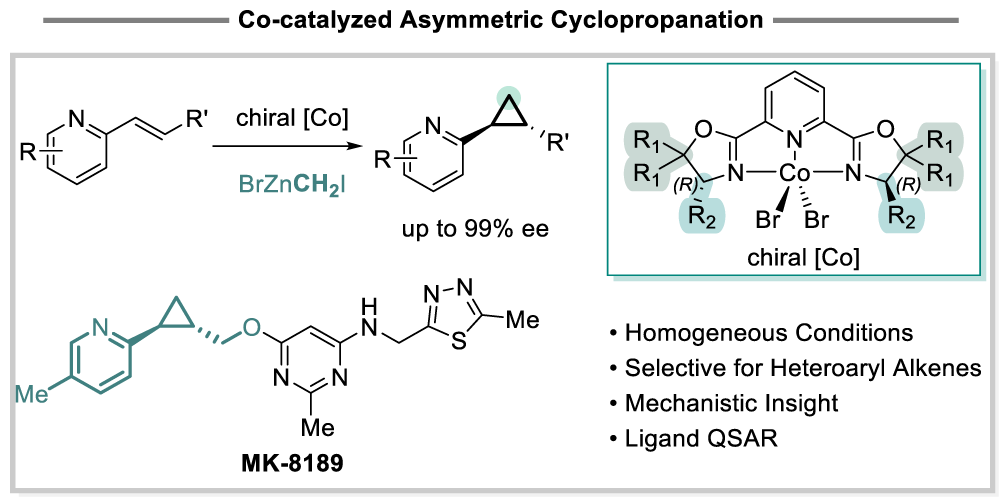
64. Cobalt-Catalyzed Asymmetric Cyclopropanation of Heteroaryl Alkenes with Homogeneous Zinc Carbenoids
M. C. Deem, D. Lehnherr, T. Wang, X. Wang, Y.-h. Lam, S. M. Silverman, J. A. Newman, Q. Lin, E. M. Phillips, M. Christensen, C. K. Chung, Z. Zhou,* H. Yang*
J. Am. Chem. Soc. 2026, DOI: 10.1021/jacs.6c05870 [PDF] doi: 10.1021/jacs.6c05870
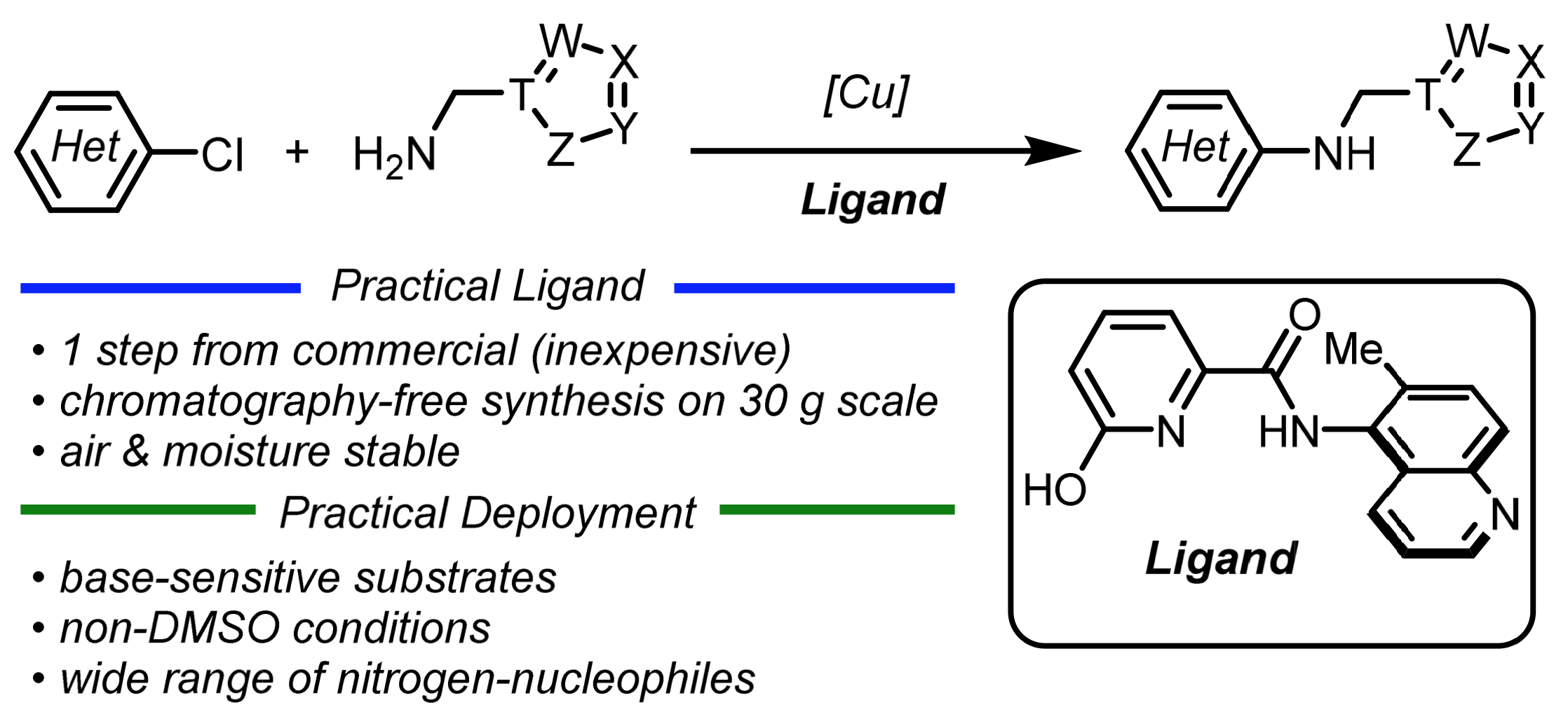
63. Development of a Practical Copper-Catalyzed C—N Cross-Coupling for Heteroaryl Chlorides and Base-Sensitive Substrates
A. McGrath,* D. Lehnherr*
ACS Catal. 2026, 16, 9333—9347. [PDF] doi: 10.1021/acscatal.6c01237
62. Asymmetric Synthesis of MK-7845, an Investigational Treatment for COVID-19
R. T. Larson, B. W. H. Turnbull,* P. J. Moon, D. Lehnherr, J. M. E. Hughes, S. Popov, G. A. Brito, T. Andreani, S. Kim, J. Herbort, R. J. Sifri, S. O. Badir, C. I. Onyeagusi, N. R. Deprez, D. Otte, D. R. Gauthier Jr., N. Kuhl, Y. Ji, A. J. Neel, R. D. Cohen, J. S. Derrick, C. Bottecchia, C. Hartmanshenn, D. A. Vargas, G. D. Poggetto, A. M. Confer, M. Zheng, Y. Qin, A. Vikram, J. A. Newman, Z. Sakhaei, R. J. Drout, L. Chen, Z. Wu, S. Castro, S. K. Betancourt, G. Longia, B. Kassim, Y. Xu, J. M. M. Dunn, N. S. Marzijarani*
Org. Lett. 2025, 27, 12505—12511. [PDF] doi: 10.1021/acs.orglett.5c03920
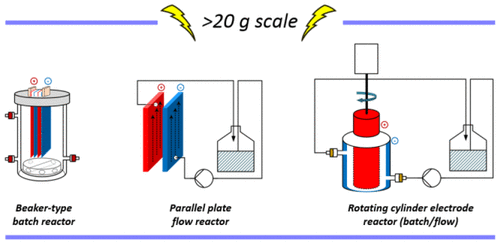
61. A Scalable Flow Electrolysis of Heterogenous Mixtures Using a Rotating Cylinder Electrode Reactor: Access to a Chiral Iminophosphorane
F. Xu, L. Chen,* D. Lehnherr,* F. Lévesque
Org. Process Res. Dev. 2025, 29, 872—880. [PDF] doi: 10.1021/acs.oprd.4c00532
60. Scaling Organic Electrosynthesis: The Crucial Interplay Between Mechanism and Mass Transport
Z. J. Oliver, D. J. Abrams, L. Cardinale, C.-J. Chen, G. L. Beutner, S. Caille, B. Cohen, L. Deng, M. Diwan, M. O. Frederick, K. Harper, J. M. Hawkins, D. Lehnherr, C. Lucky, A. Meyer, S. Noh, D. Nunez, K. Quasdorf, J. Teli, S. S. Stahl,* M. Schreirer*
ACS Cent. Sci. 2025, 11, 528—538. [PDF] doi: 10.1021/acscentsci.4c01733
59. Selective Hydrogenolysis of Organohalides
H. Yang,* D. Lehnherr*
Nat. Synth. 2025, 4, 414—416. [PDF] doi: 10.1038/s44160-024-00733-z
58. Adoption of Electrochemistry within the Pharmaceutical Industry: Insights from an Industry Wide-Survey
A. C. Ferretti,* B. Cohen, L. Deng, M. Diwan, M. O. Frederick, D. Lehnherr
Org. Process Res. Dev. 2025, 29, 322—332. [PDF] doi: 10.1021/acs.oprd.4c00353
57. Scalable Flow Electrosynthesis of Iminophosphoranes
D. Lehnherr,* L. Chen,* F. Lévesque, C. Nietupski, M. Weisel, V. Mdluli, K. Mattern, B. M. Armstrong, T. Chen, R. D. Cohen
Org. Process Res. Dev. 2025, 29, 100—113. [PDF] doi: 10.1021/acs.oprd.4c00372
56. Hydrogen Isotope Labeling of Pharmaceuticals Via Dual Hydrogen Isotope Exchange Pathways Using CdS Quantum Dot Photocatalyst
R. Maity, O. Dungan, F. A. Perras, J. Li, D. Liu, S. Ren, D. Lehnherr, Z. Huang, E. M. Phillips, M. Adeyemo, J. Frimpong, T. Quainoo, Z.-F. Liu, L. Luo*
J. Am. Chem. Soc. 2024, 146, 34141—34151. [PDF] doi: 10.1021/jacs.4c13857
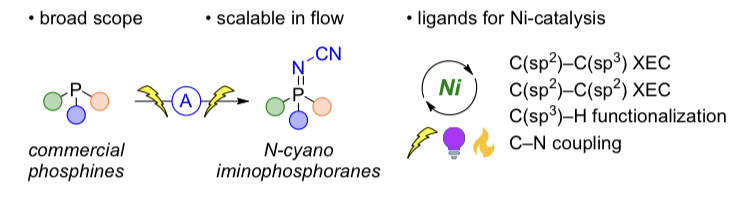
55. Electrosynthesis of Iminophosphoranes and Applications in Nickel Catalysis
V. Mdluli, D. Lehnherr,* Y.-h. Lam,* M. T. Chaudhry, J. A. Newman, J. O. DaSilva, E. L. Regalado
Chem. Sci. 2024, 15, 5980—5992. [PDF] doi: 10.1039/D3SC05357A
54. Overview of Recent Scale-Ups in Organic Electrosynthesis (2000—2023)
D. Lehnherr,* L. Chen*
Org. Process Res. Dev. 2024, 28, 338—366. [PDF] doi: 10.1021/acs.oprd.3c00340
53.
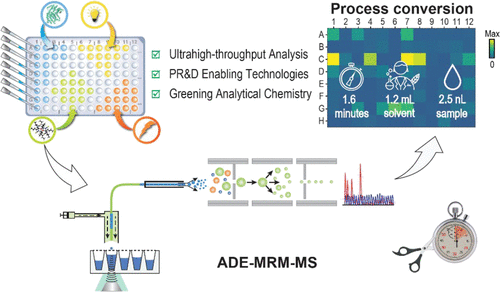
 Accelerating Pharmaceutical Process Development with an Acoustic Droplet Ejection-Multiple Reaction Monitoring-Mass Spectrometry Workflow
Accelerating Pharmaceutical Process Development with an Acoustic Droplet Ejection-Multiple Reaction Monitoring-Mass Spectrometry Workflow
H. Hu,*, A. N. Singh, D. Lehnherr, V. Mdluli, S. W. Chun, A. M. Makarewicz, J. R. Gouker, O. Ukaegbu, S. Li, X. Wen, D. G. McLaren, J. E. Velasquez, J. C. Moore, S. Galanie, E. Appiah-Amponsah, E. L. Regalado
Anal. Chem. 2024, 96, 1138—1146. [PDF] doi: 10.1021/acs.analchem.3c04211
• Highlighted on Cover
52. Multigram Electrochemical Hofmann Rearrangement using a Spinning Three-Dimensional Anode
B. K. Malviya, C. Bottecchia, K. Stone, D. Lehnherr, F. Lévesque, C. O. Kappe, D. Cantillo*
Org. Process Res. Dev. 2023, 27, 2183—2191. [PDF] doi: 10.1021/acs.oprd.3c00332
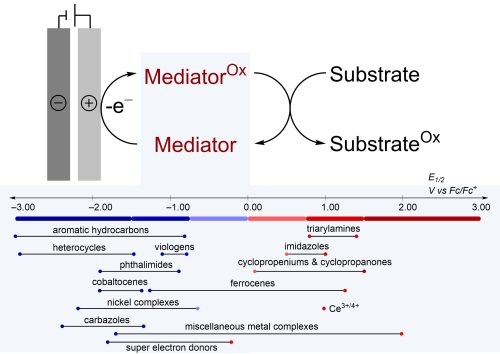
51. Overview of Outer-Sphere Electron Transfer Mediators for Electrosynthesis
L. Cardinale, S. S. Stahl,* D. Kalyani,* D. Lehnherr*
Adv. Catal. 2023, 72, 57—102. [PDF] doi: 10.1016/bs.acat.2023.07.009
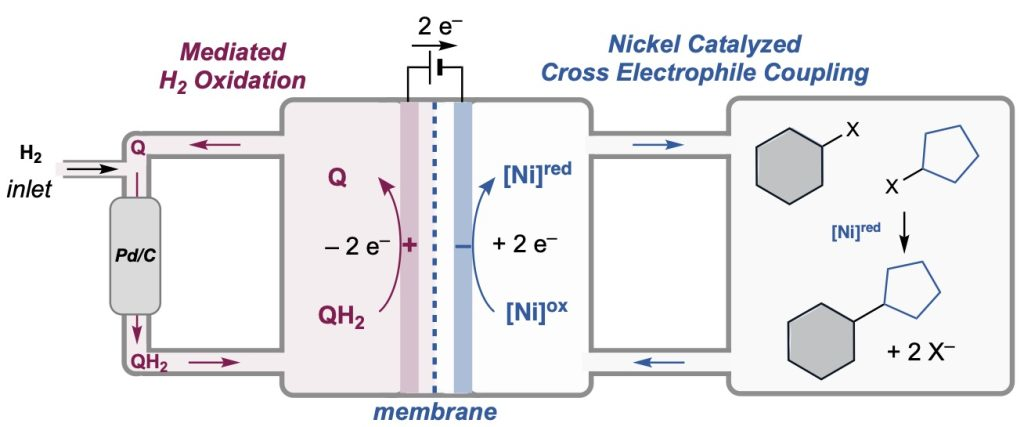
50. Quinone-Mediated Hydrogen Anode for Non-Aqueous Reductive Electrosynthesis
J. Twilton,‡ M. R. Johnson,‡ V. Sidana, M. C. Franke, C. Bottecchia, D. Lehnherr, F. Lévesque, S. M. M. Knapp, L. Wang, J. B. Gerken, C. M. Hong, T. P. Vickery, M. D. Weisel, N. A. Strotman, D. J. Weix,* T. W. Root,* S. S. Stahl* (‡ = equal contribution)
Nature 2023, 623, 71—76. [PDF] doi: 10.1038/s41586-023-06534-2
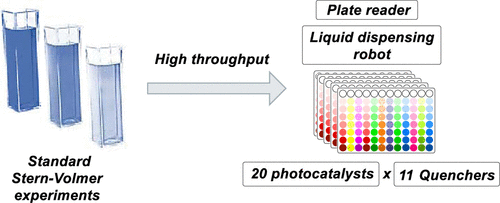
49. High Throughput Determination of Stern Volmer Quenching Constants for Common Photocatalysts and Quenchers
R. Motz, A. C. Sun, D. Lehnherr, S. Ruccolo*
ACS Org. Inorg. Au 2023, 3, 266—273. [PDF] doi: 10.1021/acsorginorgau.3c00019
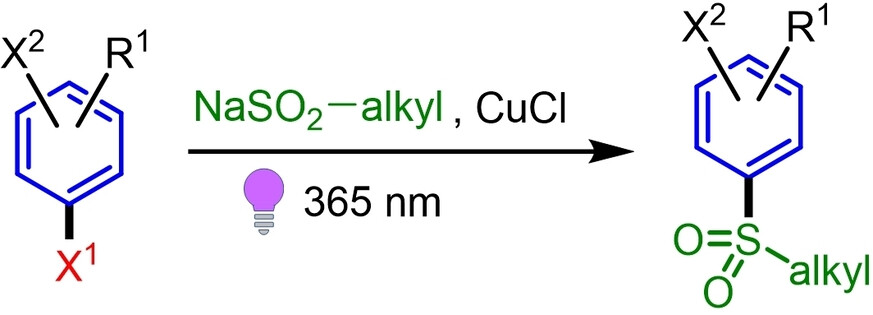
48.
 Copper-Enabled Photo-Sulfonylation of Aryl Halides Using Alkylsulfinates
Copper-Enabled Photo-Sulfonylation of Aryl Halides Using Alkylsulfinates
V. Mdluli, D. Lehnherr,* Y.-h. Lam,* Y. Ji, J. Newman, J. Kim
Adv. Synth. Catal. 2023, 26, 3876—3886. [PDF] doi: 10.1002/adsc.202300257
• Highlighted on Cover
• Highlighted in Synform
47. Emerging reaction technologies in pharmaceutical development: Challenges and opportunities in electrochemistry, photochemistry, and biocatalysis
B. Cohen*, D. Lehnherr, M. Sezen-Edmonds, J. H. Forstater, M. O. Frederick, L. Deng, A. C. Ferretti, K. Harper, M. Diwan
Chem. Eng. Res. Des. 2023, 192, 622—637. [PDF] doi: 10.1016/j.cherd.2023.02.050
46. Industrial Applications of Asymmetric Oxidations
V. Mdluli, D. Lehnherr*
In Comprehensive Chirality, 2nd ed.; Cossy, J., Ed.; Academic Press, 2024, 206—240. [PDF] doi: 10.1016/B978-0-32-390644-9.00032-9
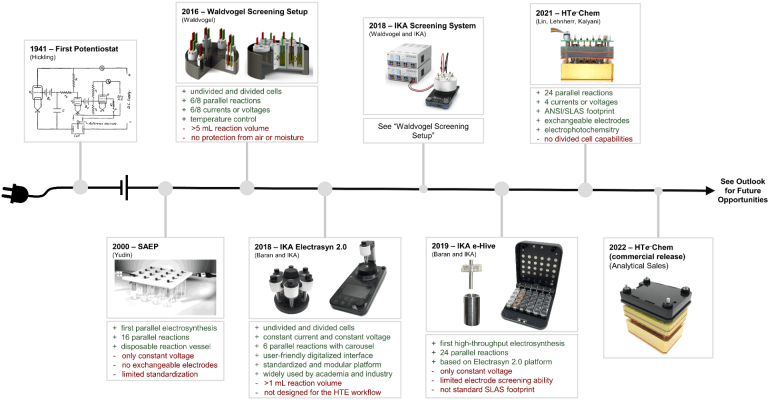
45. High-Throughput Experimentation for Electrochemistry
J. Rein, S. Lin,* D. Kalyani,* D. Lehnherr*
ACS Symposium Series, Vol. 1419, 2022, 167—187. [PDF] doi: 10.1021/bk-2022-1419.ch010
In The Power of High-Throughput Experimentation: General Topics and Enabling Technologies for Synthesis and Catalysis (Volume 1); M. H. Emmert, M. Jouffroy, D. C. Leitch, Eds.; ACS Symposium Series, Vol. 1419, American Chemical Society, 2022.
eBook ISBN: 9780841297562. Hardcover ISBN13: 9780841297579.
44. Pentacenones as Divergent Intermediates to Unsymmetrically Substituted Pentacenes: Synthesis and Crystallographic Analysis
Z. W. Schroeder, R. McDonald, M. J. Ferguson, W. A. Chalifoux, R. R. Tykwinski, D. Lehnherr*
J. Org. Chem. 2022, 87, 16236—16249. [PDF] doi: 10.1021/acs.joc.2c01755
43. Kilo-Scale Electrochemical Oxidation of a Thioether to a Sulfone: A Workflow for Scaling Up Electrosynthesis
C. Bottecchia,* D. Lehnherr,* F. Lévesque, M. Reibarkh, Y. Ji, V. L. Rodrigues, H. Wang, Y.-h. Lam, T. P. Vickery, B. M. Armstrong, K. A. Mattern, K. Stone, M. K. Wismer, A. N. Singh, E. L. Regalado, K. M. Maloney, N. A. Strotman
Org. Process Res. Dev. 2022, 26, 2423—2437. [PDF] doi: 10.1021/acs.oprd.2c00111
• Selected as ACS Editors' Choice (open acess for a limited time)
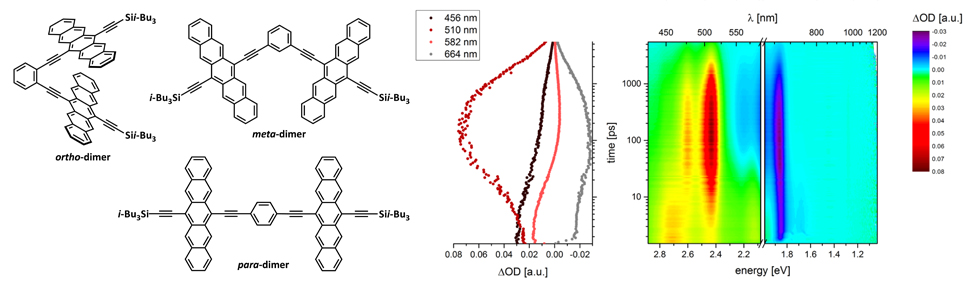
42. Parallel versus Twisted Pentacenes: Conformational Impact on Singlet Fission
I. Papadopoulos, S. R. Reddy, P. B. Coto*, D. Lehnherr, D. Thiel, M. Thoss, R. R. Tykwinski, D. M. Guldi*
J. Phys. Chem. Lett. 2022, 13, 5094—5100. [PDF] doi: 10.1021/acs.jpclett.2c01395
41. Efficient Aliphatic Hydrogen-Isotope Exchange with Tritium Gas through the Merger of Photoredox and Hydrogenation Catalysts
H. Yang,* Z. Huang, D. Lehnherr,* Y.-h. Lam, S. Ren, N. A. Strotman
J. Am. Chem. Soc. 2022, 144, 5010—5022. [PDF] doi: 10.1021/jacs.1c13265
40. Electrochemically Driven Cross-Electrophile Coupling of Alkyl Halides
W. Zhang, L. Lu, W. Zhang, Y. Wang, S. D. Ware, J. Mondragon, J. Rein, N. Strotman, D. Lehnherr, K. A. See,* S. Lin*
Nature 2022, 604, 292—297. [PDF] doi: 10.1038/s41586-022-04540-4
• Highlighted in Synform
39. Photons or Electrons? A Critical Comparison of Electrochemistry and Photoredox Catalysis for Organic Synthesis
N. E. S. Tay, D. Lehnherr,* T. Rovis*
Chem. Rev. 2022, 122, 2487—2649. [PDF] doi: 10.1021/acs.chemrev.1c00384
• Part of special issue on "Photochemical Catalytic Processes".
38. Manufacturing Process Development for Belzutifan, Part 2: A Continuous Flow Visible Light-Induced Benzylic Bromination
C. Bottecchia,‡,* F. Lévesque,‡,* J. P. McMullen, Y. Ji, M. Reibarkh, F. Peng, L. Tan, G. Spencer, J. Nappi, D. Lehnherr, K. Narsimhan, M. K. Wismer, L. Chen, Y. Lin, S. M. Dalby (‡ = equal contribution)
Org. Process Res. Dev. 2022, 26, 516—524. [PDF] doi: 10.1021/acs.oprd.1c00240
37. Benzylic Photobromination for the Synthesis of Belzutifan: Elucidation of Reaction Mechanisms Using In Situ LED-NMR
Y. Ji,* C. Bottecchia, F. Lévesque, K. Narsimhan, D. Lehnherr, J. P. McMullen, S. M. Dalby, K.-J. Xiao, M. Reibarkh*
J. Org. Chem. 2022, 87, 2055—2062. [PDF] [Supporting Information] doi: 10.1021/acs.joc.1c01465
36. Unlocking the Potential of High-Throughput Experimentation for Electrochemistry with a Standardized Microscale Reactor
J. Rein, J. R. Annand, M. K. Wismer, J. Fu, J. C. Siu, A. Klapars, N. A. Strotman, D. Kalyani,* D. Lehnherr,* S. Lin*
ACS Cent. Sci. 2021, 7, 1347—1355. [PDF] [Supporting Information] doi: 10.1021/acscentsci.1c00328
• HTe—Chem is now available from Analytical Sales.
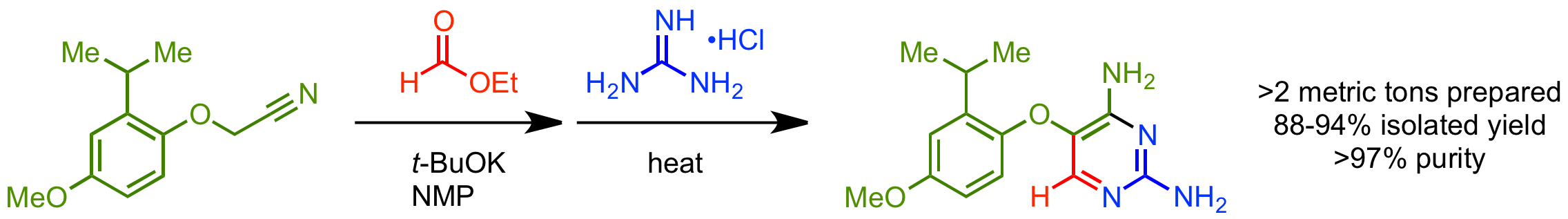
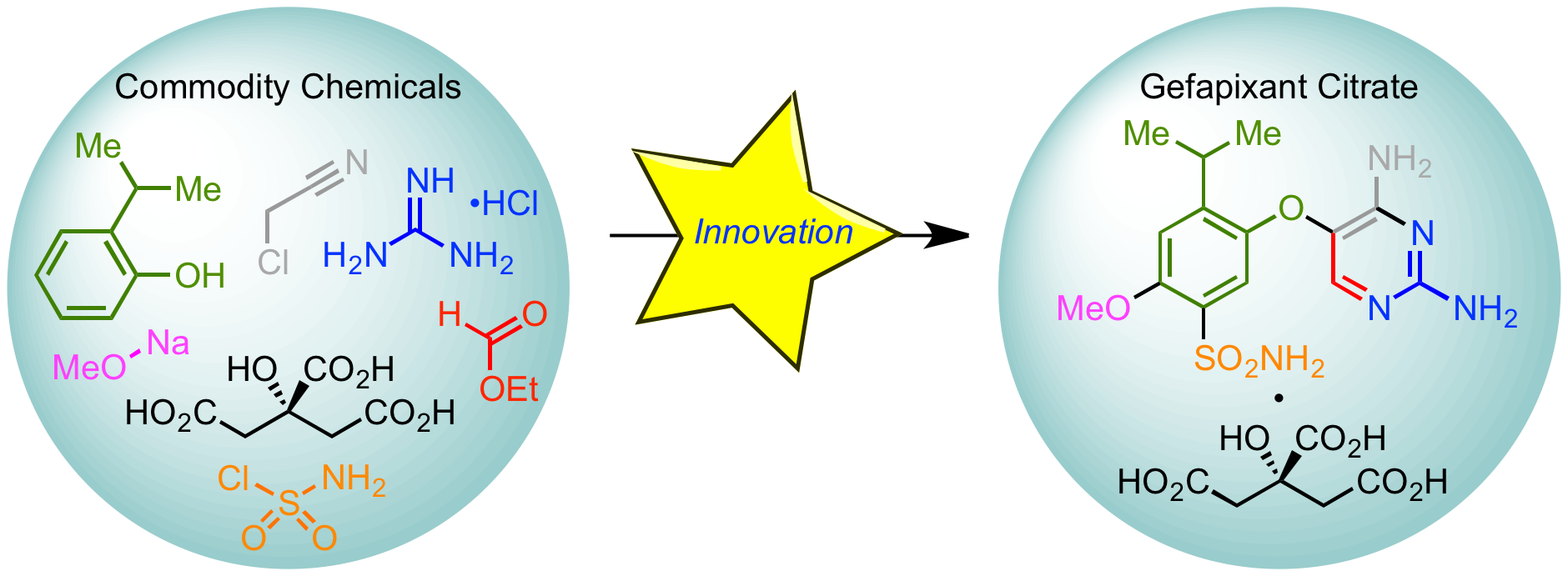
35.  Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
(MK-7264) Part 3: Development of a One-Pot Formylation-Cyclization Sequence to the Diaminopyrimidine Core
K. Basu,* D. Lehnherr,* G. E. Martin, R. A. Desmond, Y.-h. Lam, F. Peng, J. Y. L. Chung, R. Arvary, M. A. Zompa, S.-W. Zhang, J. Liu, Z. E. X. Dance, P. Larpent, R. D. Cohen, F. J. Guzman, N. J. Rogus, M. J. Di Maso, H. Ren, K. M. Maloney
Org. Process Res. Dev. 2020, 24, 2462—2477. [PDF] [Supporting Information] doi: 10.1021/acs.oprd.0c00246
• Featured on Issue Cover
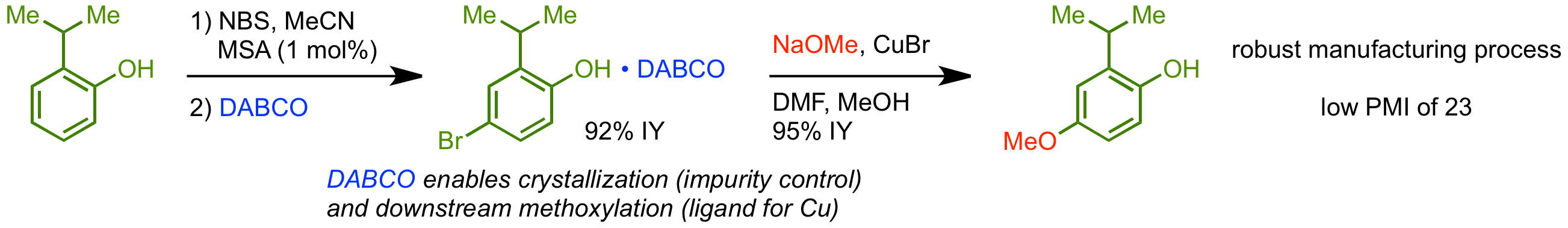
34.  Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
(MK-7264) Part 2: Development of a Robust Process for Phenol Synthesis.
F. Peng,* G. R. Humphrey, K. Maloney, D. Lehnherr, M. Weisel, F. Lévesque, J. R. Naber, A. P. J. Brunskill, P. Larpent, S.-W. Zhang, A. Y. Lee, R. A. Arvary, C. H. Lee, D. Bishara, K. Narsimhan, E. Sirota, M. Whittington.
Org. Process Res. Dev. 2020, 24, 2453—2461. [PDF] [Supporting Information] doi: 10.1021/acs.oprd.0c00241
• Featured on Issue Cover
33.  Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
Development of a Green & Sustainable Manufacturing Process for Gefapixant Citrate
(MK-7264) Part 1: Introduction and Process Overview.
H. Ren,* K. M. Maloney,* K. Basu, M. J. Di Maso, G. R. Humphrey, F. Peng, R. Desmond, D. A. L. Otte, E. Alwdi, W. Liu, S.-W. Zhang, S. Song, R. A. Arvary, M. A. Zompa, D. Lehnherr, G. E. Martin, H. Yao, D. Chang, A. E. Mohan, F. J. Guzman, L. Jellett, A. Y. Lee, G. Spencer, E. S. Fisher, J. R. Naber, H. Gao, S. Lohani, R. T. Ruck, L.-C. Campeau
Org. Process Res. Dev. 2020, 24, 2445—2452. [PDF] [Supporting Information] doi: 10.1021/acs.oprd.0c00248
• Featured on Issue Cover
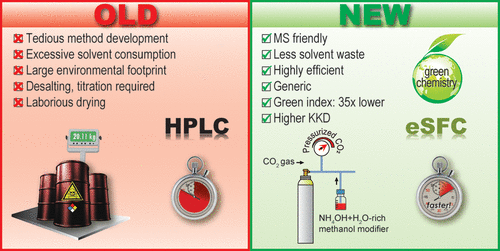
32. Generic Enhanced Sub/Supercritical Fluid Chromatography: The Blueprint for Highly Productive and Sustainable Separation of Primary Hindered Amines
J. O. DaSilva,* D. Lehnherr, J. Liu, R. Bennett, I. A. H. Ahmad, M. Hicks, B. F. Mann, D. A. DiRocco, E. L. Regalado*
ACS Sustainable Chem. Eng. 2020, 8, 6011—6021. [PDF] [Supporting Information] doi: 10.1021/acssuschemeng.0c00777
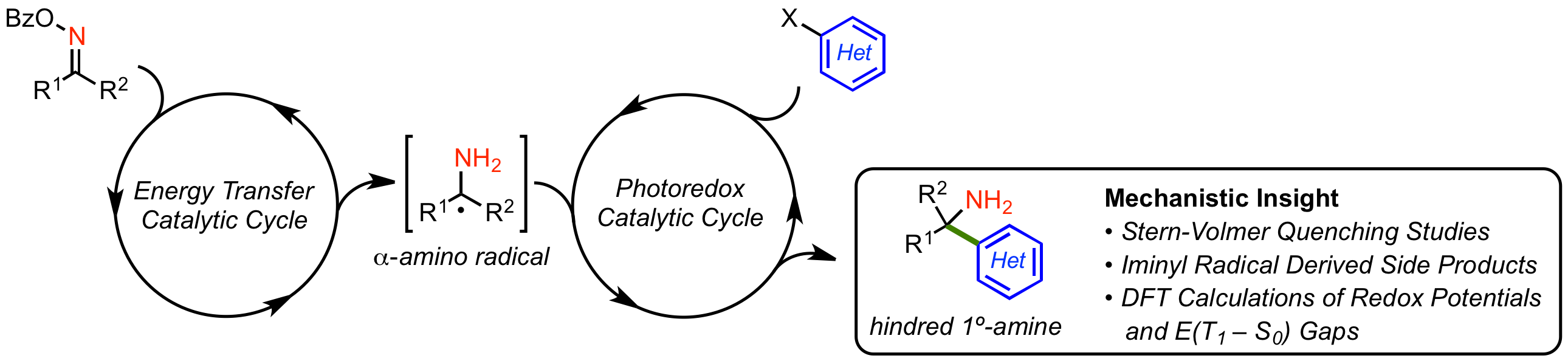
31. Synthesis of Sterically Hindered Primary Amines by Concurrent Tandem Photoredox Catalysis
M. C. Nicastri, D. Lehnherr,* Y.-h. Lam, D. A. DiRocco, T. Rovis*
J. Am. Chem. Soc. 2020, 142, 987—998. [PDF] [Supporting Information] doi: 10.1021/jacs.9b10871
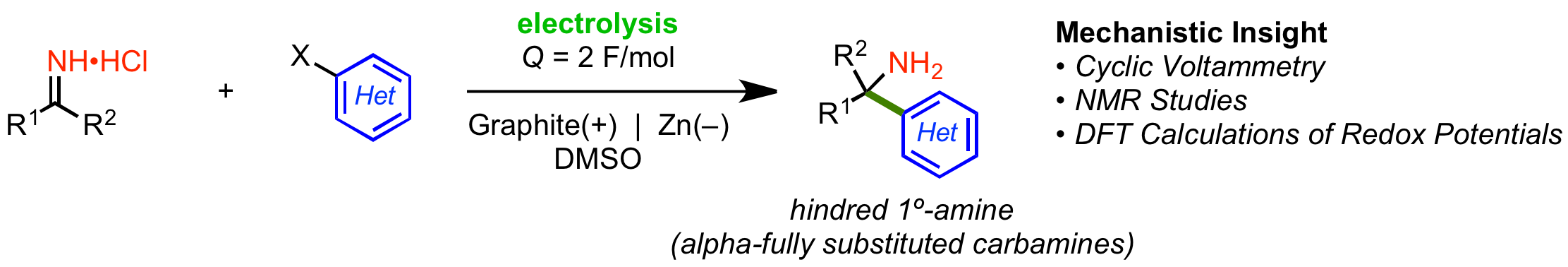
30. Electrochemical Synthesis of Hindered Primary and Secondary Amines via Proton-Coupled Electron Transfer
D. Lehnherr,* Y.-h. Lam, M. C. Nicastri, J. Liu, J. A. Newman, E. L. Regalado, D. A. DiRocco, T. Rovis*
J. Am. Chem. Soc. 2020, 142, 468—478. [PDF] [Supporting Information] doi:10.1021/jacs.9b10870
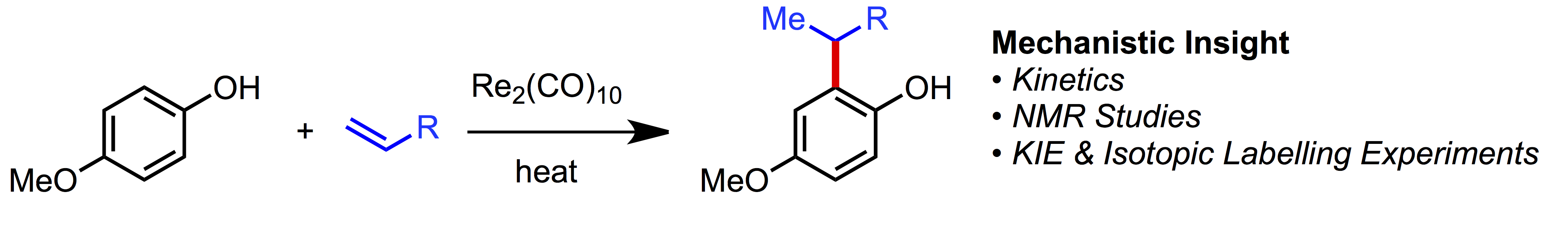
29. Mechanistic Study of a Re-Catalyzed Monoalkylation of Phenols
D. Lehnherr,* X. Wang,* F. Peng, M. Weisel, M. Reibarkh, K. M. Maloney
Organometallics 2019, 38, 103—118. [PDF] [Supporting Information] doi: 10.1021/acs.organomet.8b00543
• Part of special issue on "The Roles of Organometallic Chemistry in Pharmaceutical Research and Development".
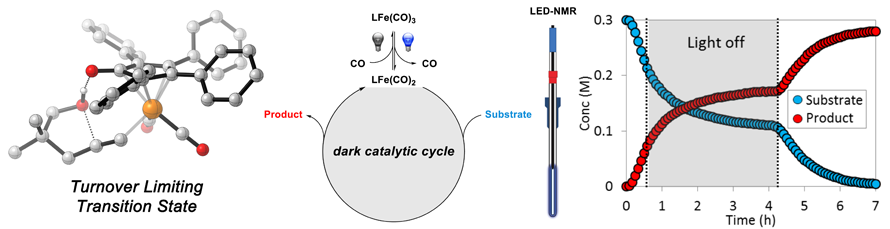
28. Discovery of a Photoinduced Dark Catalytic Cycle Using in Situ LED-NMR Spectroscopy
D. Lehnherr,‡,* Y. Ji,‡,* A. J. Neel, R. D. Cohen, A. P. J. Brunskill, J. Yang, M. Reibarkh
(‡ = equal contribution)
J. Am. Chem. Sci. 2018, 140, 13843—13853. [PDF] [Supporting Information] doi: 10.1021/jacs.8b08596
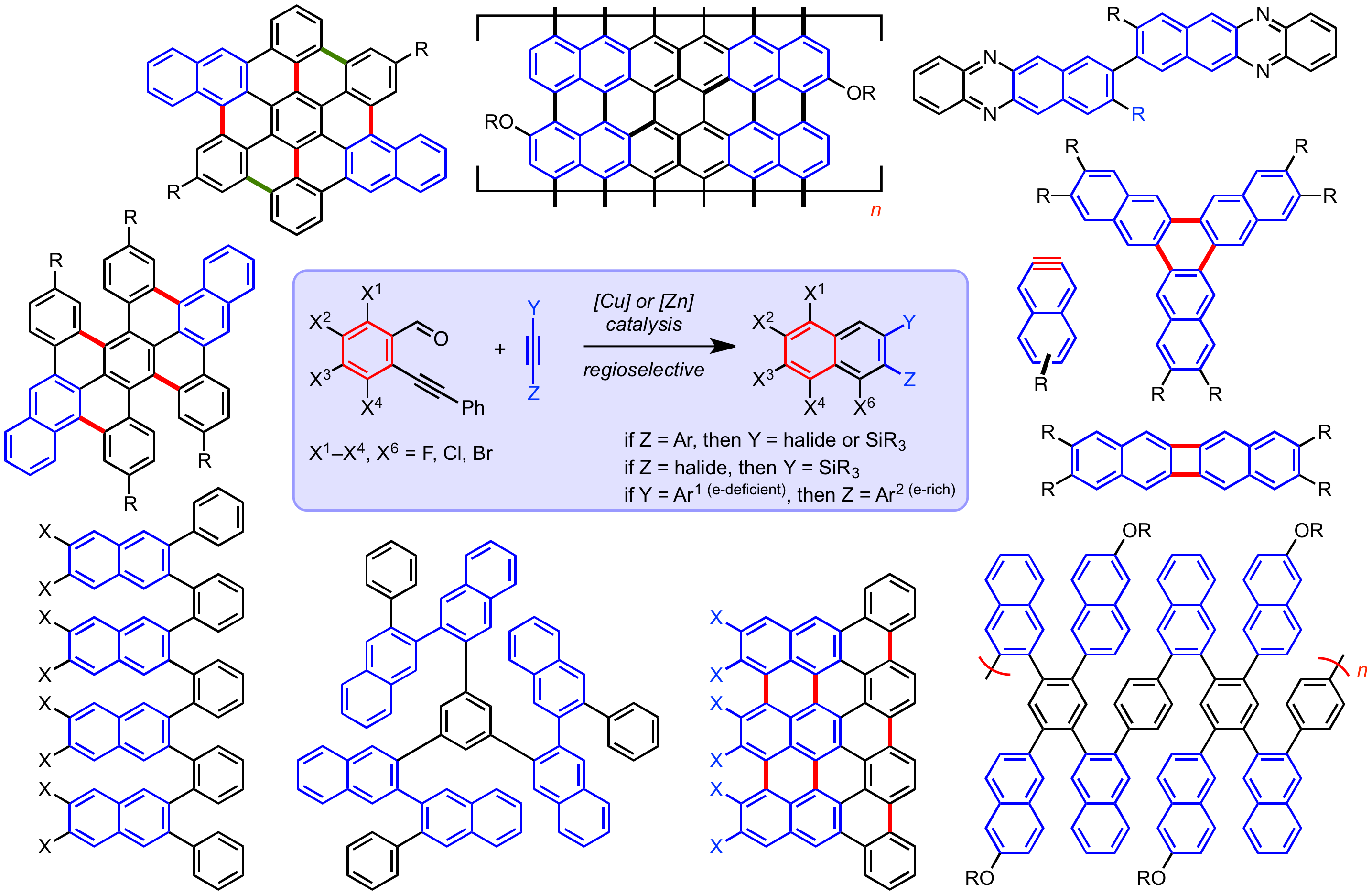
27. Alkyne Benzannulation Reactions for the Synthesis of Novel Aromatic Architectures
S. J. Hein, D. Lehnherr, H. Arslan, F. J. Uribe-Romo, W. R. Dichtel*
Acc. Chem. Res. 2017, 50, 2776—2788. [PDF] doi: 10.1021/acs.accounts.7b00385
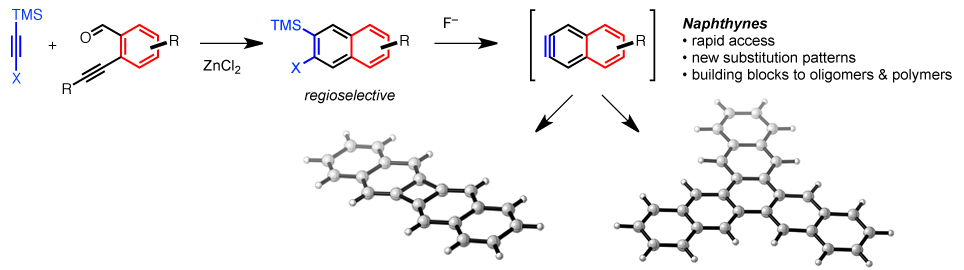
26. Rapid Access to Substituted 2-Naphthyne Intermediates via the Benzannulation of Halogenated Silylacetylenes
S. J. Hein, D. Lehnherr, W. R. Dichtel*
Chem. Sci. 2017, 8, 5675—5681. [PDF] [Supporting Information] doi: 10.1039/c7sc01625e
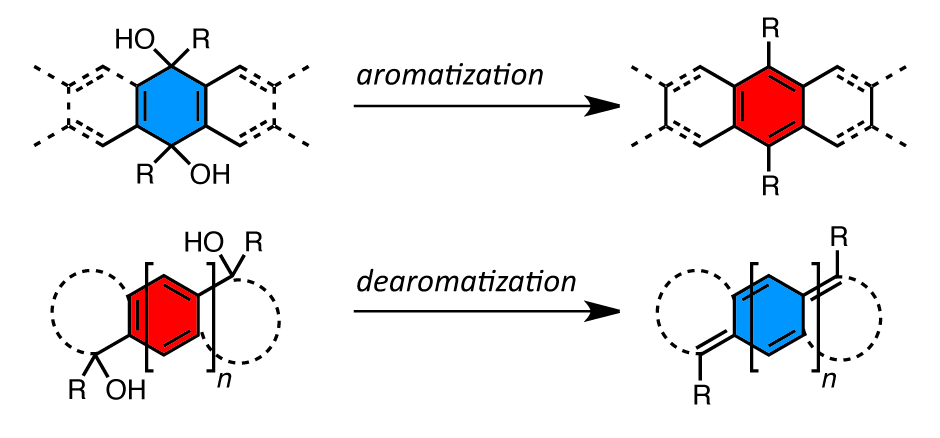
25. Reductive Aromatization/Dearomatization and Elimination Reactions to Access Conjugated Polycyclic Hydrocarbons, Heteroacenes, and Cumulenes
J. L. Marshall, D. Lehnherr, B. D. Lindner, R. R. Tykwinski*
ChemPlusChem 2017, 82, 967—1001. [PDF] doi: 10.1002/cplu.201700168
• Part of special issue on "Novel Aromatics: From Synthesis to Applications".
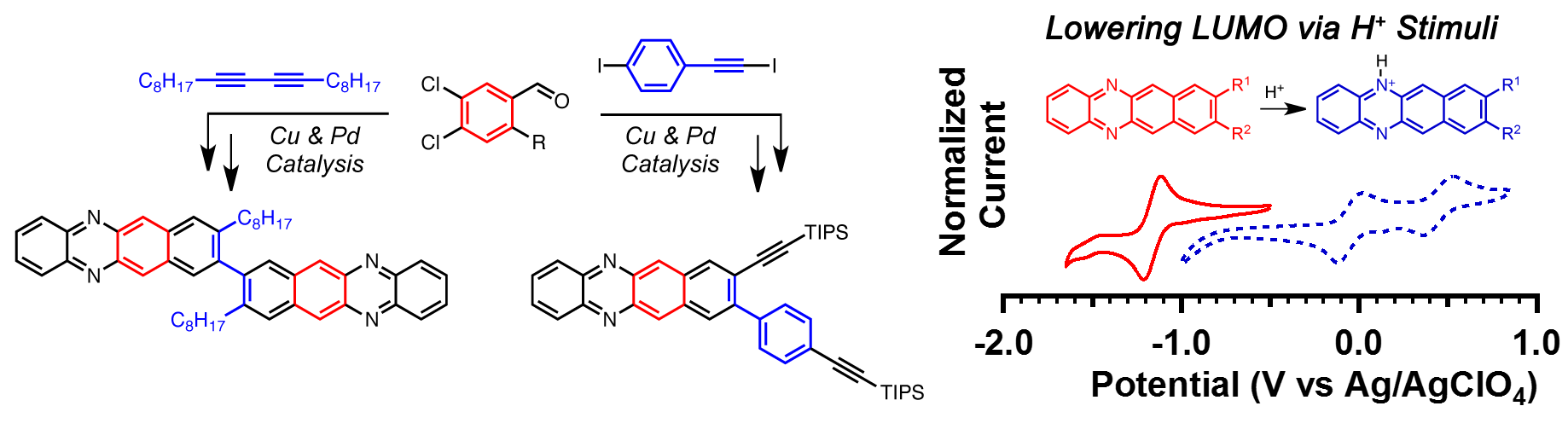
24. Diazatetracenes Derived From the Benzannulation of Acetylenes: Electronic Tuning via Substituent Effects and External Stimuli
D. Lehnherr, J. M. Alzola, C. R. Mulzer, S. J. Hein, W. R. Dichtel*
J. Org. Chem. 2017, 82, 2004—2010. [PDF] [Supporting Information] doi: 10.1021/acs.joc.6b02840
• Highlighted in Synfacts
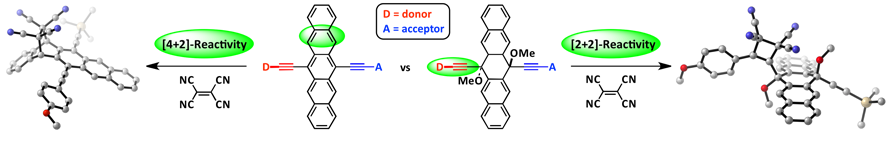
23. Synthesis, Physical Properties, and Chemistry of Donor/Acceptor Substituted Pentacenes
D. Lehnherr, M. Adam, A. H. Murray, R. McDonald, F. Hampel, R. R. Tykwinski*
Can. J. Chem. 2017, 95, 303—314. [PDF] [Supporting Information] doi: 10.1139/cjc-2016-0450
• Part of special issue dedicated to Prof. Reginald Mitchell.
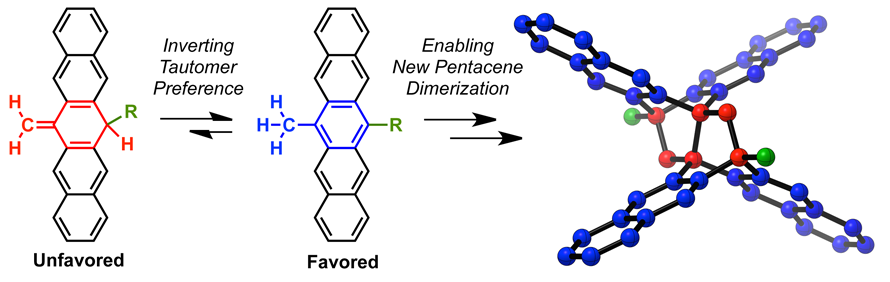
22. Tautomerization and Dimerization of 6,13-Disubstituted Derivatives of Pentacene
M. Garcia-Borràs, A. Konishi, A. Waterloo, Y. Liang, Y. Cao, C. Hetzer, D. Lehnherr, F. Hampel, K. N. Houk,* R. R. Tykwinski*
Chem. Eur. J. 2017, 23, 6111—6117. [PDF] [Supporting Information] doi: 10.1002/chem.201604099
• Part of special issue on "Cooperative Effects in Chemistry".
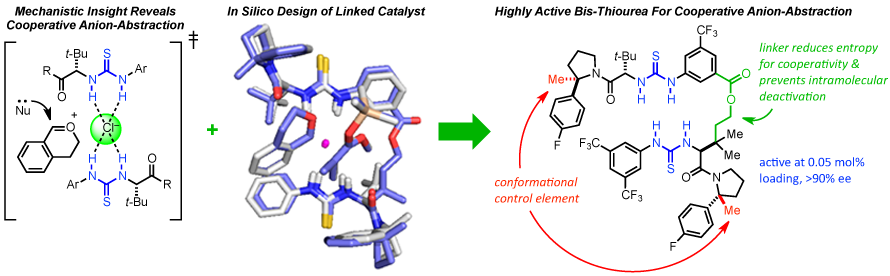
21. Mechanism-Guided Development of Highly Active Bis-Thiourea Catalysts for Cooperative Anion-Abstraction Catalysis
C. R. Kennedy,‡ D. Lehnherr,‡ N. S. Rajapaksa, D. D. Ford, Y. Park, E. N. Jacobsen*
(‡ = equal contribution)
J. Am. Chem. Soc. 2016, 138, 18122—18127. [PDF] [Supporting Information] doi: 10.1002/jacs.6b09205
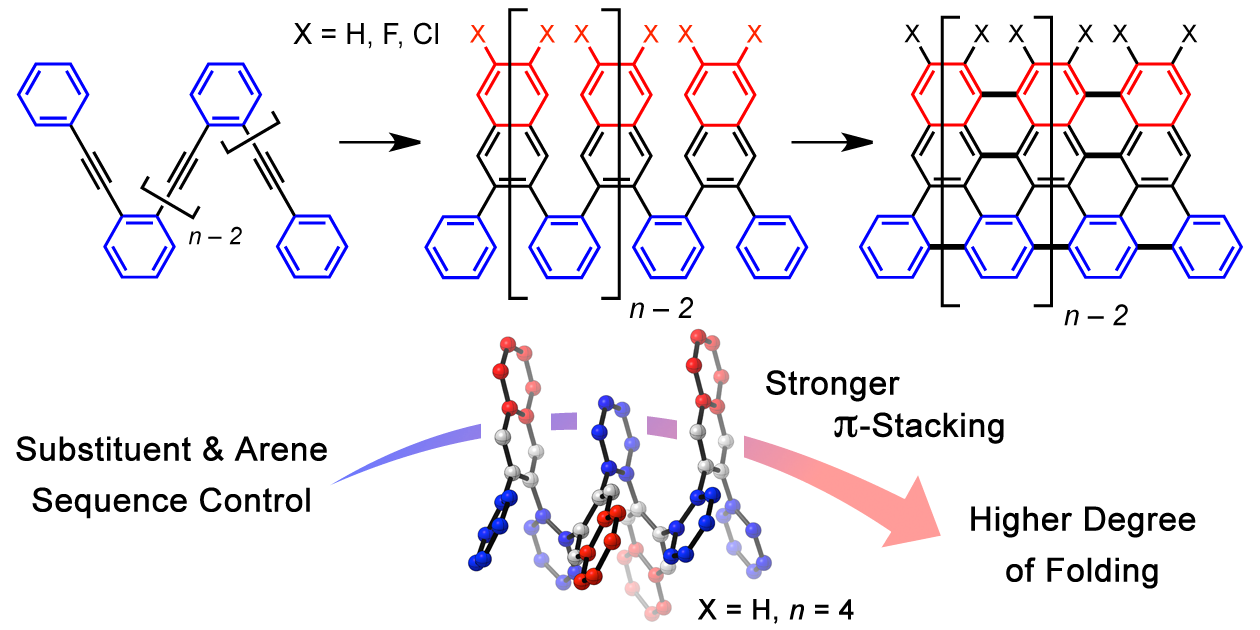
20. Sequence-Defined Oligo(ortho-Arylene) Foldamers Derived from the Benzannulation of ortho(Arylene ethynylene)s
D. Lehnherr, C. Chen, Z. Pedramrazi, C. R. DeBlase, J. M. Alzola, I. Keresztes, E. B. Lobkovsky, M. F. Crommie,* W. R. Dichtel*
Chem. Sci. 2016, 7, 6357—6364. [PDF] [Supporting Information] doi: 10.1039/C6SC02520J
• Highlighted in Synfacts
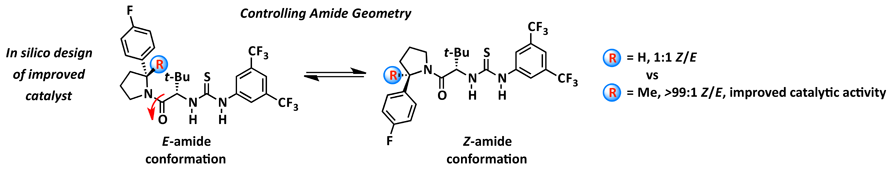
19. Conformational Control of Chiral Amido Thiourea Catalysts Enables Improved Catalytic Activity and Enantioselectivity
D. Lehnherr, D. D. Ford, A. Bendelsmith, C. R. Kennedy, E. N. Jacobsen*
Org. Lett. 2016, 18, 3214—3217. [PDF] [Supporting Information] doi: 10.1021/acs.orglett.6b01435h
• Highlighted in Synfacts
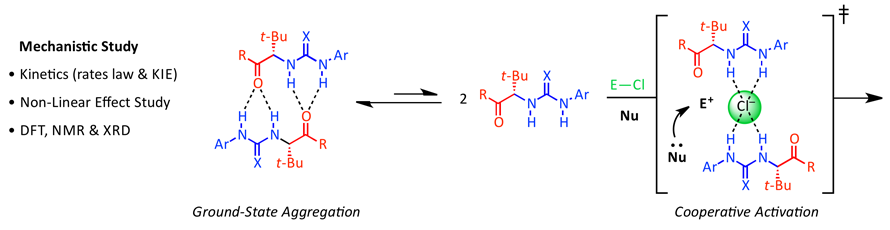
18. Anion-Abstraction Catalysis: The Cooperative Mechanism of α-Chloroether Activation by Dual H-Bond Donors
D. D. Ford, D. Lehnherr, C. R. Kennedy, E. N. Jacobsen*
ACS Catal. 2016, 6, 4616—4620. [PDF] [Supporting Information] doi: 10.1021/acscatal.6b01384
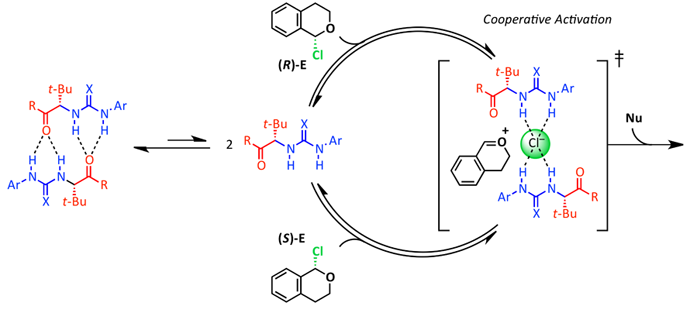
17. On- and Off-Cycle Catalyst Cooperativity in Anion-Binding Catalysis
D. D. Ford,‡ D. Lehnherr,‡ C. R. Kennedy, E. N. Jacobsen*
(‡ = equal contribution)
J. Am. Chem. Soc. 2016, 138, 7860—7863. [PDF] [Supporting Information] doi: 10.1021/6b04686
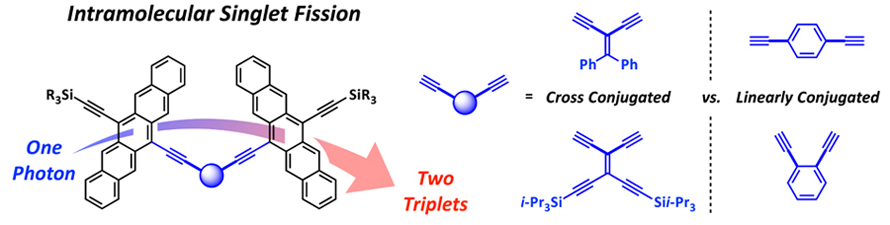
16. Solution-Based Intramolecular Singlet Fission in Cross-Conjugated Pentacene Dimers
J. Zirzlmeier, R. Casillas, S. R. Reddy, P. B. Coto, D. Lehnherr, E. T. Chernick, I. Papadopoulos, M. Thoss*, R. R. Tykwinski*, D. M. Guldi*
Nanoscale 2016, 8, 10113—10123. [PDF] [Supporting Information] doi: 10.1039/C6NR02493a
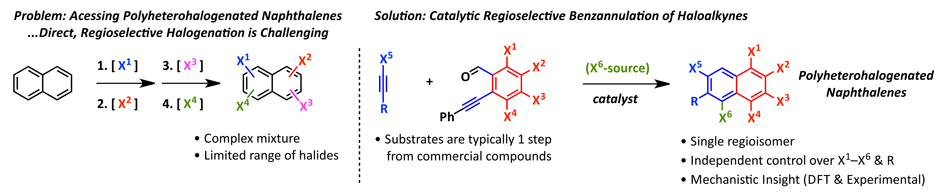
15.  Regioselective Synthesis of Polyheterohalogenated Naphthalenes via the Benzannulation
Regioselective Synthesis of Polyheterohalogenated Naphthalenes via the Benzannulation
of Haloalkynes
D. Lehnherr, J. M. Alzola, E. B. Lobkovsky, W. R. Dichtel*
Chem. Eur. J. 2015, 21, 18122—18127. [PDF] [Supporting Information] doi: 10.1002/chem.2001503418
• Featured on Issue Cover
14. Singlet Fission in Pentacene Dimers
J. Zirzlmeier, D. Lehnherr, P. B. Coto, E. T. Chernick, R. Casillas, B. Basel, M. Thoss*, R. R. Tykwinski*, D. M. Guldi*
Proc. Natl. Acad. Sci. U.S.A. 2015, 112, 5325—5330 [PDF] [Supporting Information] doi: 10.1073/pnas.1422436112
13. Aryl Substitution of Pentacenes
A. R. Waterloo, A.-C. Sale, D. Lehnherr, F. Hampel, R. R. Tykwinski*
Beilstein J. Org. Chem. 2014, 10, 1692—1705 [PDF] [Supporting Information] doi: 10.3762/bjoc.10.178
• Part of special issue on “Functionalized Carbon-Nanomaterials” (Guest Editor: Prof. Anke Krueger).
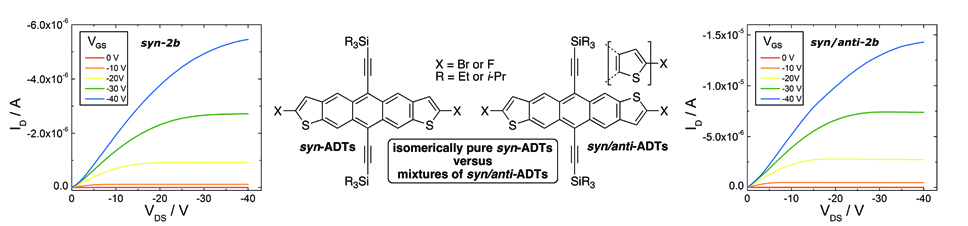
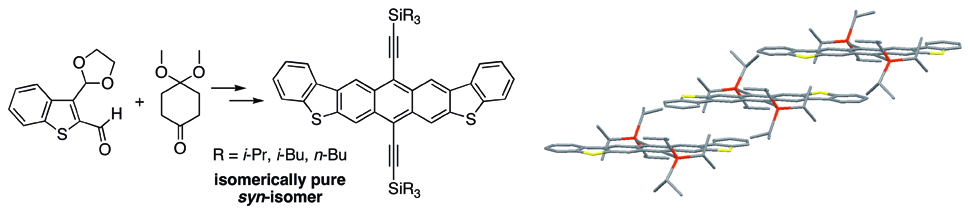
12. Isomerically Pure syn-Anthradithiophenes: Synthesis, Properties and FET Performance
D. Lehnherr, A. Waterloo, K. P. Goetz, M. M. Payne, F. Hampel, J. E. Anthony,* O. D. Jurchescu,* R. R. Tykwinski*
Org. Lett. 2012, 14, 3660—3663 [PDF] [Supporting Information] doi: 10.1021/ol301503k
11. Synthesis and Properties of Isomerically Pure Anthrabisbenzothiophenes
D. Lehnherr, R. Hallani, R. McDonald, J. E. Anthony,* R. R. Tykwinski*
Org. Lett. 2012, 14, 62—65 [PDF] [Supporting Information] doi: 10.1021/ol202843x
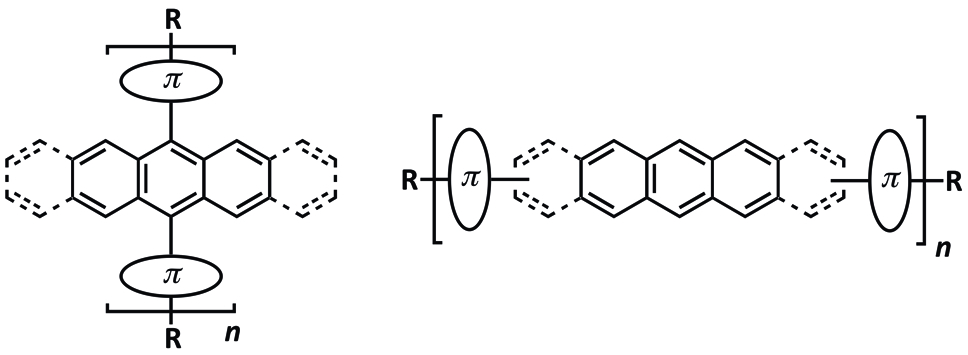
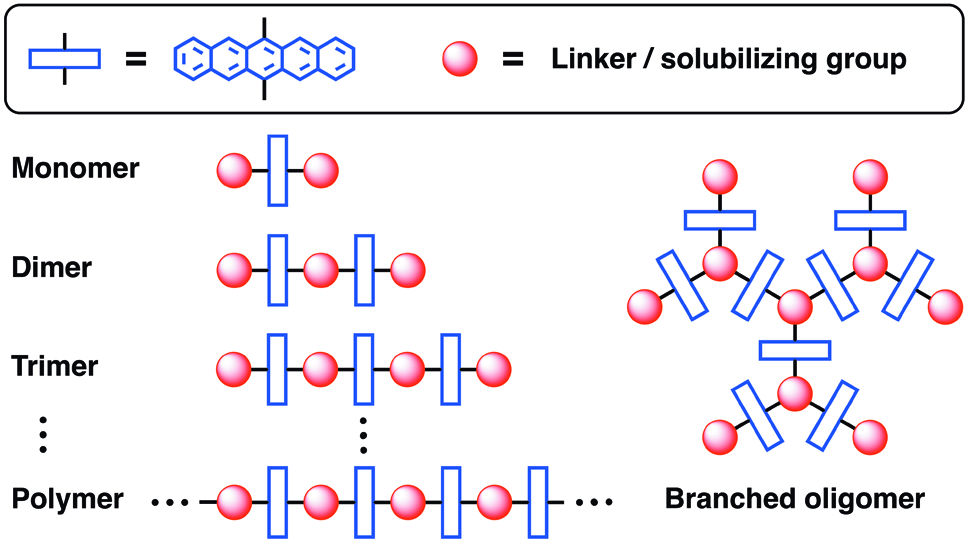
10. Oligomers and Polymers Based on Anthracene, Tetracene, Pentacene, Naphthodithiophene, and Anthradithiophene Building Blocks
D. Lehnherr, R. R. Tykwinski*
Aust. J. Chem. 2011, 64, 919—929 [PDF] doi: 10.1071/CH11169
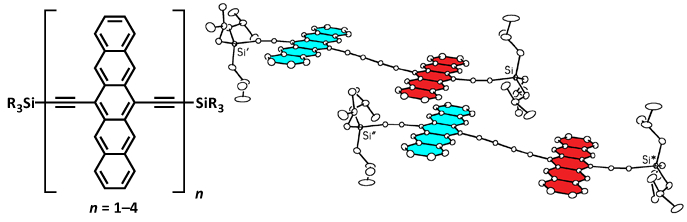
9. A Modular Synthetic Approach to Conjugated Pentacene Di-, Tri-, and Tetramers
D. Lehnherr, A. H. Murray, R. McDonald, R. R. Tykwinski*
Angew. Chem. Int. Ed. 2010, 49, 6190—6194 [PDF] [Supporting Information] doi: 10.1002/anie.201000555
8. Oligomers and Polymers Based on Pentacene Building Blocks
D. Lehnherr, R. R. Tykwinski*
Materials 2010, 3, 2772—2800 [PDF] doi: 10.3390/ma3042772
• Review as part of the special issue on “Conjugated Oligomers” (Guest Editor: Prof. Herbert Meier).
7. Pentacene-Based Polycyclic Aromatic Hydrocarbon Dyads with Cofacial Solid-State Pi-Stacking
D. Lehnherr, A. H. Murray, R. McDonald, M. J. Ferguson, R. R. Tykwinski*
Chem. Eur. J. 2009, 15, 12580—12584 [PDF] [Supporting Information] doi: 10.1002/chem.200902179
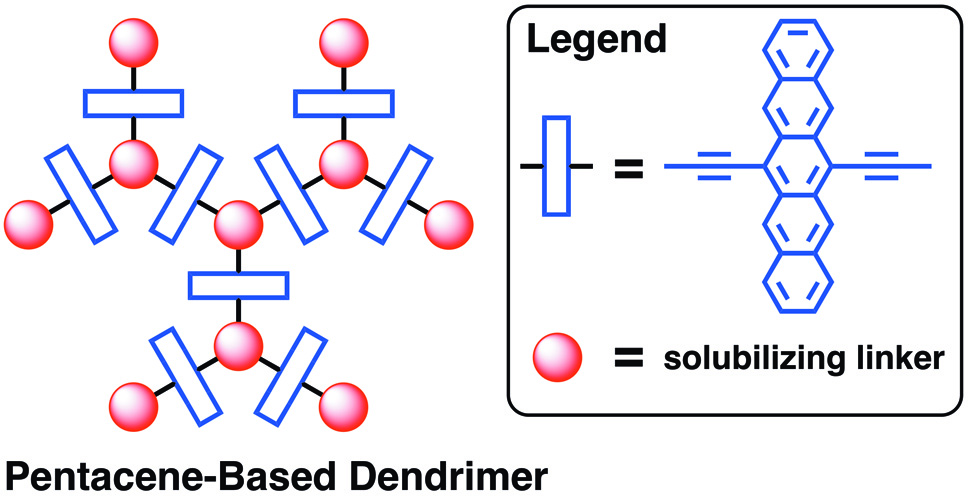
6. Pentacene Dendrimers: Synthesis and Thin Film Photoconductivity Measurements of Branched Pentacene Oligomers
D. Lehnherr, J. Gao, F. A. Hegmann,* R. R. Tykwinski*
J. Org. Chem. 2009, 74, 5017—5024 [PDF] [Supporting Information] doi: 10.1021/jo9007089
5. Synthesis of Soluble Oligo- and Polymeric Pentacene-Based Materials
D. Lehnherr, R. McDonald, M. J. Ferguson, R. R. Tykwinski*
Tetrahedron 2008, 64, 11449—11461 [PDF] [Supporting Information] doi: 10.1016/j.tet.2008.09.041
• Part of “The Synthesis of Carbon-Rich Nanomolecules” symposia in print.
4. Synthesis and Electronic Properties of Conjugated Pentacene Dimers
D. Lehnherr, J. Gao, F. A. Hegmann,* R. R. Tykwinski*
Org. Lett. 2008, 10, 4779—4782 [PDF] [Supporting Information] doi: 10.1021/ol801886h
3. Exploring Electronically Polarized Pentacenes
D. Lehnherr, R. McDonald, R. R. Tykwinski*
Org. Lett. 2008, 10, 4163—4166 [PDF] [Supporting Information] doi: 10.1021/ol801464k
2. Pentacene Oligomers and Polymers: Functionalization of Pentacene To Afford Mono-, Di-, Tri-, and Polymeric Materials
D. Lehnherr, R. R. Tykwinski*
Org. Lett. 2007, 9, 4583—4586 [PDF] [Supporting Information] doi: 10.1021/ol702094d
1. Formal Intramolecular Photoredox Chemistry of Meta-Substituted Benzophenones
D. Mitchell, M. Lukeman, D. Lehnherr, P. Wan*
Org. Lett. 2005, 7, 3387—3389 [PDF] [Supporting Information] doi: 10.1021/ol051381u
View Citation Metrics by Google Scholar. |
Current h-index: 34. |